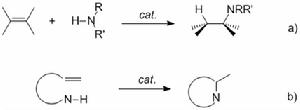
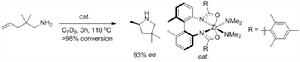
雖然催化劑的中心金屬的研究範圍很廣,但是總的來講,過渡金屬所體現的催化性能要比其他主族金屬要好一些。稀土金屬催化劑的研究覆蓋了早期氫胺化反應的研究。目前來看,稀土配合物不對稱催化分子內烯烴氫胺化反應已經取得一些的突破,Marks小組對於這方面反應作了不少系統的工作,研究範圍涵蓋了從機理到套用。
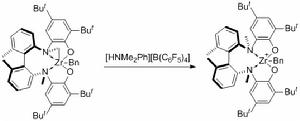
相比前過渡金屬,後過渡金屬通常具備更加優越的官能團容忍性,但是它們往往需要酸性條件和氮保護的
胺,更為不利的一點是他們比較低的催化效率和催化劑壽命。Widenhoefer小組成功實現了胺基活化的胺分子內對烯烴及聯烯的氫胺化反應。儘管最近Hartwig小組也取得了重要突破,對部分非活化分子內烯胺實現了有效的催化反應,但是底物限制還是非常大,對於非活化烯胺底物也尚未有不對稱催化的報導。
[1] (a) T. E. Müller, K. C. Hultzsch, M. Yus, F. Foubelo, M. Tada, Chem. Rev. 2008, 108, 3795–3892. (b) Odom, A. L. Dalton Trans. 2005, 225. (c) Hultzsch, K. C. AdV. Synth. Catal. 2005, 347, 367. (d) Hultzsch, K. C.; Gribkov, D. V.; Hampel, F. J. Organomet. Chem. 2005, 690, 4441. (e) Hong, S.; Marks, T. J. Acc. Chem. Res. 2004, 37, 673. (f) Doye, S. Synlett 2004, 1653. (g) Beller, M.; Tillack, A.; Seayad, J. In Transition Metals for Organic Synthesis, 2nd ed.; Beller, M., Bolm, C., Eds.; Wiley-VCH: Weinheim, 2004; pp 91. (h) Roesky, P. W.; Mueller, T. E. Angew. Chem., Int. Ed. 2003, 42, 2708. (i) Pohlki, F.; Doye, S. Chem. Soc. Rev. 2003, 32, 104. (j) Bytschkov, I.; Doye, S. Eur. J. Org. Chem. 2003, 68, 935. (k) Seayad, J.; Tillack, A.; Hartung, C. G.; Beller, M. Adv. Synth. Catal. 2002, 344, 795. (l) Togni, A. In Catalytic Heterofunctionalization; Togni, A., Gruetzmacher, H., Eds.; Wiley-VCH: New York, 2001; pp 91. (m) Nobis, M.; Drieâen-Holscher, B. Angew. Chem., Int. Ed. 2001, 40, 3983. (n) Eisen, M. S.; Straub, T.; Haskel, A. J. Alloys Compd. 1998, 271, 116. (n) Mueller, T. E.; Beller, M. Chem. Rev. 1998, 98, 675.
[2] (a)Gribkov, D. V.; Hultzsch, K. C. Angew. Chem., Int. Ed. 2004, 43, 5542. (c) Thomson, R. K.; Bexrud, J. A.; Schafer, L. L. Organometallics 2006, 25, 4069. (d) Wood, M. C.; Leitch, D. C.; Yeung, C. S.; Kozak, J. A.; Schafer, L. L. Angew. Chem., Int. Ed. 2007, 46, 354.
[3] Zhang Z., Bender C. F., Widenhoefer R. A. J. Am. Chem. Soc. 2007, 129, 14148.
[4] Liu Z., Hartwig J. F. J. Am. Chem. Soc. 2008, 130, 1570.
[5] (a) Bytschkov, I.; Doye, S. Eur. J. Org. Chem. 2003, 2003, 935. (b) Gribkov, D. V.; Hultzsch, K. C. Angew. Chem., Int. Ed. 2004, 43, 5542. (c) Knight P. D., Munslow I., O’Shaughnessy P. N. , Scott P. Chem. Commun. 2004, 894 (d) Thomson, R. K.; Bexrud, J. A.; Schafer, L. L. Organometallics 2006, 25, 4069.
